|
Toll Free: (877) 796-6397 -- USA and Canada only -- |
Fax: +1-832-582-8590 Orders: +1-832-582-8158 |
Tech Support: +1-832-582-8158 Ext:3 Please provide your Order Number in the email. |
Technical Data
| Formula | C14H19BCl2N2O4 |
|||
| Molecular Weight | 361.03 | CAS No. | 1072833-77-2 | |
| Solubility (25°C)* | In vitro | DMSO | 72 mg/mL (199.42 mM) | |
| Ethanol | 72 mg/mL (199.42 mM) | |||
| Water | Insoluble | |||
|
* <1 mg/ml means slightly soluble or insoluble. * Please note that Selleck tests the solubility of all compounds in-house, and the actual solubility may differ slightly from published values. This is normal and is due to slight batch-to-batch variations. * Room temperature shipping (Stability testing shows this product can be shipped without any cooling measures.) |
||||
Preparing Stock Solutions
Biological Activity
| Description | Ixazomib (MLN2238) inhibits the chymotrypsin-like proteolytic (β5) site of the 20S proteasome with IC50 and Ki of 3.4 nM and 0.93 nM in cell-free assays, respectively, also inhibits the caspase-like (β1) and trypsin-like (β2) proteolytic sites, with IC50 of 31 and 3500 nM. Ixazomib (MLN2238) induces autophagy. Phase 3. | ||||
|---|---|---|---|---|---|
| Targets |
|
||||
| In vitro | At higher concentrations, MLN2238 also inhibits the caspase-like (β1) and trypsin-like (β2) proteolytic sites with IC50 of 31nM and 3.5uM, respectively. MLN2238 inhibits Calu-6 cell with IC50 of 9.7 nM. MLN2238 is a selective, potent, and reversible inhibitor of the proteasome in tumor cells. MLN2238 shows time-dependent reversible proteasome inhibition. Both MLN2238 and Bortezomib shows time-dependent reversible proteasome inhibition; however, the proteasome dissociation half-life for MLN2238 is determined to be ∼6-fold faster than that of Bortezomib (18 and 110 minutes, respectively). MLN2238 dissociates more rapidly from the proteasome than Bortezomib, consistent with faster recovery of proteasome activity observed in the Proteasome-Glo assay. MLN2238 has a greater overall tumor pharmacodynamic effect than Bortezomib as assessed by 20S inhibition. [1]MLN2238 is the biologically active form of MLN9708. [2] | ||||
| In vivo | MLN2238 induces a greater pharmacodynamic response than Bortezomib in xenograft tumors. MLN2238 shows greater maximum and sustained tumor proteasome inhibition compared with Bortezomib in xenograft models. These results confirm that the improved tumor exposure seen with MLN2238 translates into an improved tumor pharmacodynamic response both at the level of and downstream from the proteasome. MLN2238 shows antitumor activity in the CWR22 xenograft model. MLN2238 shows greater tumor pharmacodynamic responses in WSU-DLCL2 xenografts compared with Bortezomib. Similarly, Bortezomib treatment only led to a minor increase in GADD34 levels in WSU-DLCL2 xenograft tumors, whereas MLN2238 strongly induces its expression. [1] MLN2238 has an improved pharmacodynamic profile and antitumor activity compared with Bortezomib in both OCI-Ly10 and PHTX22L models. [2] | ||||
| Features | A first-in-class proteasome inhibitor that has improved pharmacokinetics (PK), pharmacodynamics(PD), and antitumor activity in preclinical studies. |
Protocol (from reference)
| Kinase Assay:[1] |
|
|---|---|
| Cell Assay:[1] |
|
| Animal Study:[2] |
|
References
|
Customer Product Validation
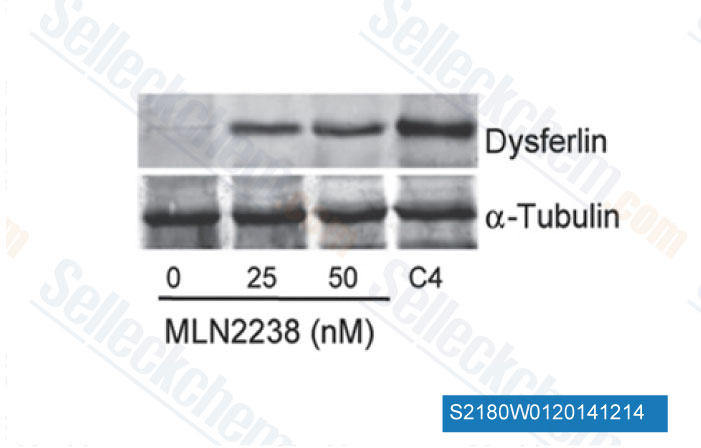
-
Data from [Data independently produced by Sci Transl Med, 2014, 6(250), 250ra112]
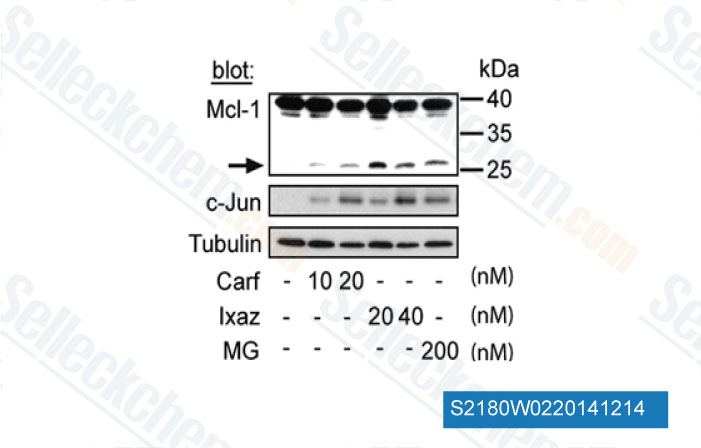
-
Data from [Data independently produced by Cancer Lett, 2014, 343(2), 286-94]
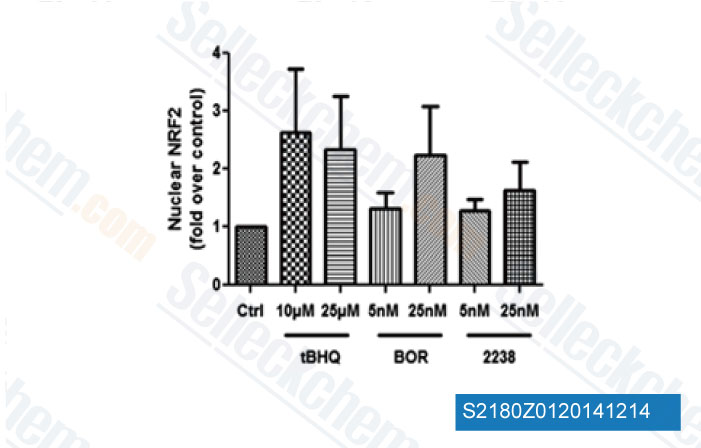
-
Data from [Data independently produced by Hemoglobin, 2014, 38(3), 188-95]
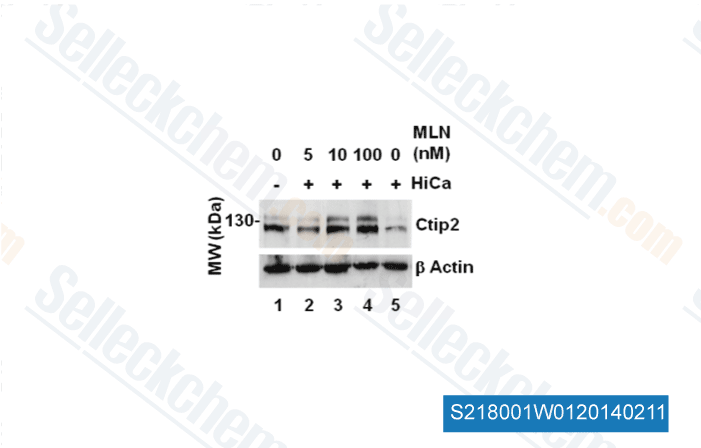
-
Data from [J Cell Sci, 2012, 125(Pt 23), 5733-44]
Selleck's Ixazomib (MLN2238) has been cited by 71 publications
| A combinatorial therapeutic approach to enhance FLT3-ITD AML treatment [ Cell Rep Med, 2023, 10.1016/j.xcrm.2023.101286] | PubMed: 37951217 |
| Targeting ITGB4/SOX2-driven lung cancer stem cells using proteasome inhibitors [ iScience, 2023, 26(8):107302] | PubMed: 37554452 |
| Targeting ITGB4/SOX2-driven lung cancer stem cells using proteasome inhibitors [ iScience, 2023, 26(8):107302] | PubMed: 37554452 |
| Dual inhibition of HSF1 and DYRK2 impedes cancer progression [ Biosci Rep, 2023, 43(1)BSR20222102] | PubMed: 36622366 |
| Proteasome inhibition as a therapeutic target for the fungal pathogen Cryptococcus neoformans [ Microbiol Spectr, 2023, 11(5):e0190423] | PubMed: 37750732 |
| Proteasome inhibition as a therapeutic target for the fungal pathogen Cryptococcus neoformans [ Microbiol Spectr, 2023, 10.1128/spectrum.01904-23] | PubMed: 37750732 |
| S100A8/S100A9 Promote Progression of Multiple Myeloma via Expansion of Megakaryocytes [ Cancer Res Commun, 2023, 3(3):420-430] | PubMed: 36923707 |
| Vactosertib, a novel TGF-β1 type I receptor kinase inhibitor, improves T-cell fitness: a single-arm, phase 1b trial in relapsed/refractory multiple myeloma [ Res Sq, 2023, rs.3.rs-3112163] | PubMed: 37503043 |
| Proteogenomics refines the molecular classification of chronic lymphocytic leukemia [ Nat Commun, 2022, 13(1):6226] | PubMed: 36266272 |
| Proteogenomics refines the molecular classification of chronic lymphocytic leukemia [ Nat Commun, 2022, 13(1):6226] | PubMed: 36266272 |
RETURN POLICY
Selleck Chemical’s Unconditional Return Policy ensures a smooth online shopping experience for our customers. If you are in any way unsatisfied with your purchase, you may return any item(s) within 7 days of receiving it. In the event of product quality issues, either protocol related or product related problems, you may return any item(s) within 365 days from the original purchase date. Please follow the instructions below when returning products.
SHIPPING AND STORAGE
Selleck products are transported at room temperature. If you receive the product at room temperature, please rest assured, the Selleck Quality Inspection Department has conducted experiments to verify that the normal temperature placement of one month will not affect the biological activity of powder products. After collecting, please store the product according to the requirements described in the datasheet. Most Selleck products are stable under the recommended conditions.
NOT FOR HUMAN, VETERINARY DIAGNOSTIC OR THERAPEUTIC USE.
