|
Toll Free: (877) 796-6397 -- USA and Canada only -- |
Fax: +1-832-582-8590 Orders: +1-832-582-8158 |
Tech Support: +1-832-582-8158 Ext:3 Please provide your Order Number in the email. |
Technical Data
| Formula | C20H24N6O2S |
||||||
| Molecular Weight | 412.51 | CAS No. | 1352226-88-0 | ||||
| Solubility (25°C)* | In vitro | DMSO | 83 mg/mL (201.2 mM) | ||||
| Ethanol | 83 mg/mL (201.2 mM) | ||||||
| Water | Insoluble | ||||||
| In vivo (Add solvents to the product individually and in order) |
|
||||||
|
* <1 mg/ml means slightly soluble or insoluble. * Please note that Selleck tests the solubility of all compounds in-house, and the actual solubility may differ slightly from published values. This is normal and is due to slight batch-to-batch variations. * Room temperature shipping (Stability testing shows this product can be shipped without any cooling measures.) |
|||||||
Preparing Stock Solutions
Biological Activity
| Description | Ceralasertib (AZD6738) is an orally active, and selective ATR kinase inhibitor with IC50 of 1 nM. Phase 1/2. | ||
|---|---|---|---|
| Targets |
|
||
| In vitro | In four Kras mutant cell lines: H23, H460, A549, and H358, AZD6738 inhibits ATR kinase activity and impairs cell viability. In ATM-deficient H23 cells, AZD6738 strongly synergizes with cisplatin to induce rapid cell death. [1] In p53 or ATM defective cells, AZD6738 treatment results in replication fork stalls and accumulation of unrepaired DNA damage, resulting in cell death by mitotic catastrophe. [2] | ||
| In vivo | In nude mice bearing H460 and H23 tumors, AZD6738 (50 mg/kg, p.o.) results in tumor growth inhibition (TGI), and the the combination with cisplatin causes rapid regression of ATM-deficient H23 tumors. [1] In nude mice bearing LoVo xenografts, a combination of AZD6738 (50 mg/kg) + IR (2 Gy) avoids toxicity while still maintaining efficacy. [3] |
Protocol (from reference)
| Cell Assay:[1] |
|
|---|---|
| Animal Study: [1] |
|
References
Customer Product Validation
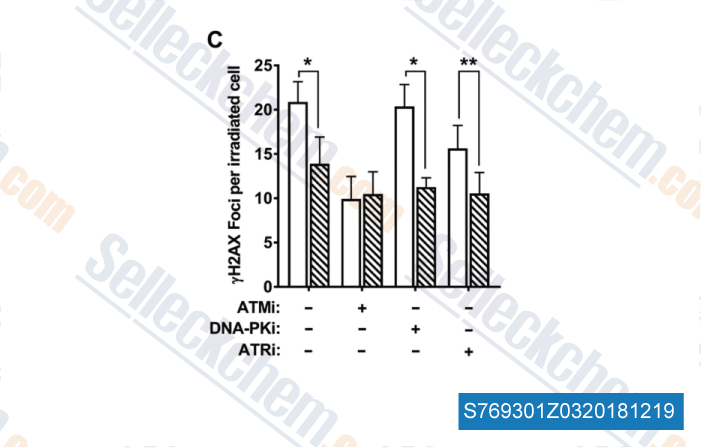
-
Data from [Data independently produced by , , Clin Cancer Res, 2018, doi:10.1158/1078-0432.CCR-18-1346]
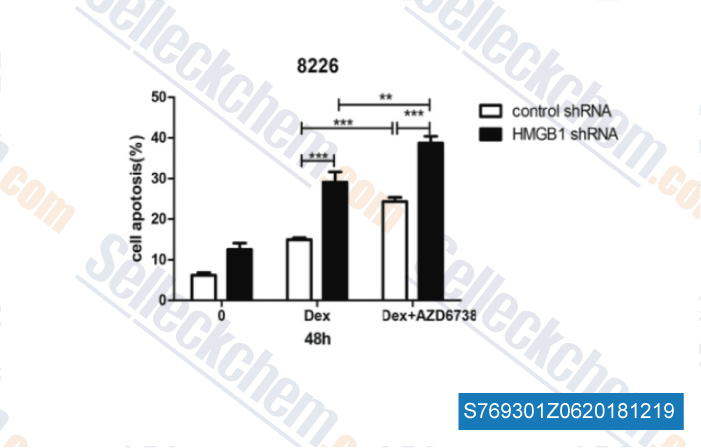
-
Data from [Data independently produced by , , J Exp Clin Cancer Res, 2018, 37(1):205]
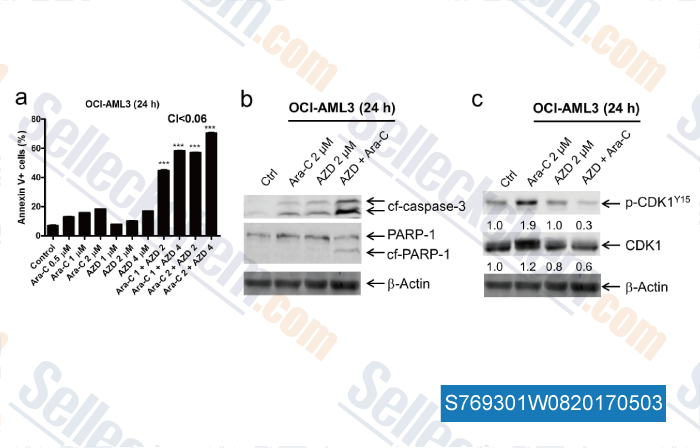
-
Data from [Data independently produced by , , Sci Rep, 2017, 7:41950]
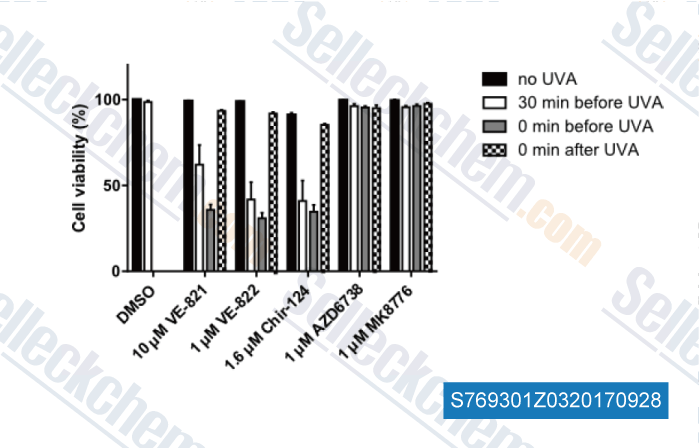
-
Data from [Data independently produced by , , J Dermatol Sci, 2016, 84(3):239-247]
Selleck's Ceralasertib (AZD6738) has been cited by 106 publications
| Functionally-instructed modifiers of response to ATR inhibition in experimental glioma [ J Exp Clin Cancer Res, 2024, 43(1):77] | PubMed: 38475864 |
| MGMT function determines the differential response of ATR inhibitors with DNA-damaging agents in glioma stem cells for GBM therapy [ Neurooncol Adv, 2024, 6(1):vdad165] | PubMed: 38213834 |
| C16orf72/HAPSTR1/TAPR1 functions with BRCA1/Senataxin to modulate replication-associated R-loops and confer resistance to PARP disruption [ Nat Commun, 2023, 14(1):5003] | PubMed: 37591890 |
| "Proteotranscriptomic analysis of advanced colorectal cancer patient derived organoids for drug sensitivity prediction" [ J Exp Clin Cancer Res, 2023, 42(1):8] | PubMed: 36604765 |
| Irreversible HER2 inhibitors overcome resistance to the RSL3 ferroptosis inducer in non-HER2 amplified luminal breast cancer [ Cell Death Dis, 2023, 14(8):532] | PubMed: 37596261 |
| DNA damage response inhibitors enhance tumour treating fields -TTFields) potency in glioma stem-like cells [ Br J Cancer, 2023, 10.1038/s41416-023-02454-0] | PubMed: 37777579 |
| Mitotic perturbation is a key mechanism of action of decitabine in myeloid tumor treatment [ Cell Rep, 2023, 42(9):113098] | PubMed: 37714156 |
| ATR protects ongoing and newly assembled DNA replication forks through distinct mechanisms [ Cell Rep, 2023, 42(7):112792] | PubMed: 37454295 |
| Effector memory T cells induce innate inflammation by triggering DNA damage and a non-canonical STING pathway in dendritic cells [ Cell Rep, 2023, S2211-1247(23)01192-0] | PubMed: 37794597 |
| Immunogenic cell death after combined treatment with radiation and ATR inhibitors is dually regulated by apoptotic caspases [ Front Immunol, 2023, 14:1138920] | PubMed: 37346039 |
RETURN POLICY
Selleck Chemical’s Unconditional Return Policy ensures a smooth online shopping experience for our customers. If you are in any way unsatisfied with your purchase, you may return any item(s) within 7 days of receiving it. In the event of product quality issues, either protocol related or product related problems, you may return any item(s) within 365 days from the original purchase date. Please follow the instructions below when returning products.
SHIPPING AND STORAGE
Selleck products are transported at room temperature. If you receive the product at room temperature, please rest assured, the Selleck Quality Inspection Department has conducted experiments to verify that the normal temperature placement of one month will not affect the biological activity of powder products. After collecting, please store the product according to the requirements described in the datasheet. Most Selleck products are stable under the recommended conditions.
NOT FOR HUMAN, VETERINARY DIAGNOSTIC OR THERAPEUTIC USE.
